|
1/12/2024 0 Comments Checkmate 649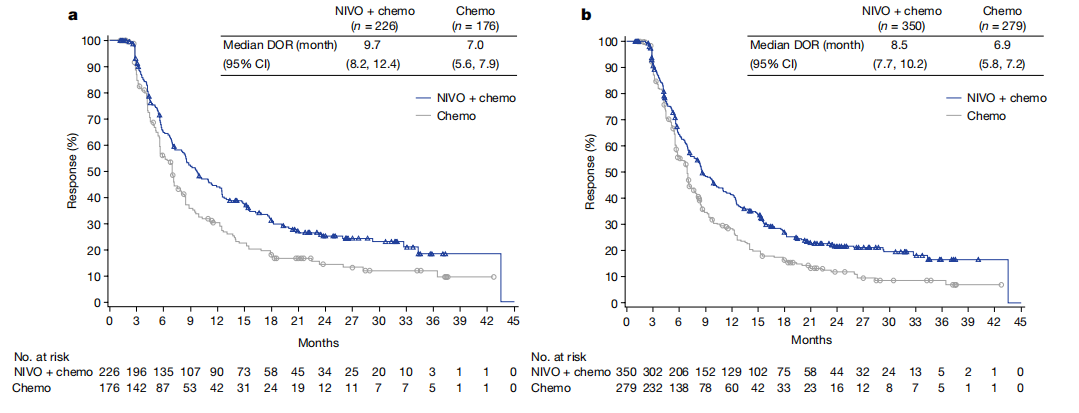 
Secondary endpoints include OS in all pts and progression-free survival and time to symptom deterioration in all pts and pts with PD-L1+ tumors. Primary endpoint is OS in pts with PD-L1+ tumors. Pts receiving chemotherapy or radiotherapy for G/GEJ cancer within the last 6 months or pts with suspected autoimmune disease, uncontrolled medical disorder, or active infection are excluded. Tumor tissue for determination of PD-L1 status must be provided from ≤ 6 months before study treatment. Methods: In this study, 870 pts aged ≥ 18 years with untreated advanced or metastatic G/GEJ cancer with or without PD-L1 expression will be randomized to receive nivo + ipi (4 doses followed by nivo monotherapy) or investigator’s choice of capecitabine/oxaliplatin (XELOX) or fluorouracil/leucovorin/oxaliplatin (FOLFOX). In Checkmate 649, serious adverse reactions occurred in 52 of patients treated with OPDIVO in combination with chemotherapy (n. This open-label, phase 3 trial will evaluate nivo + ipi as first-line therapy for pts with G/GEJ cancer (CheckMate 649 NCT02872116). Fatal adverse reactions occurred in 5 (1.6) patients who received OPDIVO in combination with YERVOY these included pneumonitis, interstitial lung disease, pulmonary embolism, and acute respiratory distress syndrome. In a phase 1/2 study in chemotherapy-refractory pts with G/GEJ/esophageal cancer with or without PD-1 ligand 1 (PD-L1) expression, second-line nivo 1 mg/kg + ipi 3 mg/kg demonstrated a manageable safety profile and resulted in 26% ORR (44% ORR in pts with PD-L1+ tumors), median OS of 6.9 months, and a 34% OS rate at 12 months (Janjigian Y, et al. Nivo, a fully human IgG4 monoclonal antibody (mAb) that targets programmed death 1 (PD-1) and ipi, a fully human IgG1 mAb that targets cytotoxic T-lymphocyte–associated protein 4, have demonstrated manageable safety profiles and efficacy in multiple tumor types and may have a synergistic effect. Therefore, new treatment options are needed to improve survival and decrease toxicity in G/GEJ cancer. This is accompanied by up to 77% grade 3/4 toxicities. Background: The combination ofoxaliplatin and fluoropyrimidine is a standard-of-care (SOC) first-line treatment of pts with metastatic G/GEJ cancer, resulting in a median overall survival (OS) of 8–11 months and objective response rate (ORR) of 30%–50%. The phase 3 CheckMate 649 trial ( Identifier: NCT02872116) randomly assigned 2031 patients with previously untreated, advanced GC, GEJC, or EAC to receive nivolumab plus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
